 Shop
ShopBroad-Spectrum CBD Softgels
Broad-Spectrum CBD Oil Tincture
Full-Spectrum CBD Softgels
Full-Spectrum CBD Oil Tincture
CBD Gummies
CBD Balm
CBD Dog Treats
All Products >

Phytocannabinoids are naturally occurring compounds found in cannabis. You're probably familiar with some of the stars like THC and CBD. What you may not know is that the raw cannabis plant does not actually contain these molecules in significant levels!
Instead, the plant material contains primarily the raw acid precursors to these popular cannabinoids. For example, in hemp or marijuana, you'll find high levels of THCA or CBDA. While research is showing promise for these 'raw' compounds, the well known 'activated' compounds are in much higher demand.
In order to convert the raw acid cannabinoids into the sought after 'activated' forms, they must undergo a process called decarboxylation.
Decarboxylation is the act of applying heat to cannabis plant material over time to create a reaction. Raw cannabinoids have a carboxyl ring or group (COOH) present in their molecular structure comprised of a carbon atom bonded to an oxygen atom (CO) and a hydroxyl group (OH). Heating removes the carbon atom from the chain and as a result carbon dioxide (CO2) is released.
When decarbed, the raw acid cannabinoids found in the plant material are converted into the sought after activated cannabinoids. This concept applies universally to all types of cannabis including marijuana and hemp. We will use both in the examples below, though we primarily focus on the hemp CBD side of things.
If you look at a cannabinoid potency lab report for raw marijuana you'll find high levels of tetrahydrocannabinolic acid (THCA), not tetrahydrocannabinol (THC). Only through decarbing is the THCA converted to THC.
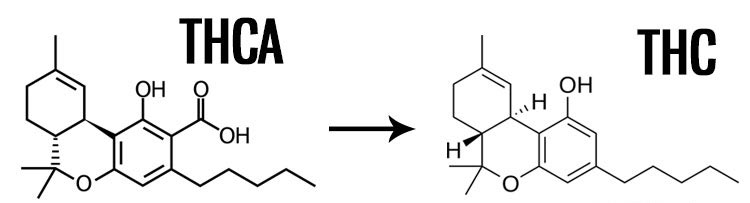
Consuming marijuana generally has a 'built-in' decarboxylation process. When smoking or vaporizing marijuana the heat rapidly decarboxylates the THCA turning it into THC just before the smoke or vapor is inhaled. For edibles, the flower used to create the product must be decarbed, often by baking in the oven, before they are added to the final product.

Decarboxylation is a concept popularly discussed in marijuana circles because it is most commonly sold as flower. While smoked hemp flower is growing in popularity, the majority of CBD products are sold as activated consumable products like tinctures and capsules.
Despite the lack of awareness, all hemp grown for extraction into CBD-rich products does need to be decarboxylated somewhere between harvest and sale as a final product.
As we cover in detail in our extraction article, CBD products are created using cannabinoid-rich oil extracts derived from hemp. When hemp is harvested for extraction, it is possible to decarboxylate it either before or after extraction.
Generally, decarboxylation before extraction is preferred because the process removes moisture which speeds the extraction process. The result is a more efficient extraction with higher yields. When using non-polar solvents in extraction methods like supercritical CO2, the activated cannabinoids are more soluble in the solvents, making them easier to separate from the plant material.
With that being said, it is possible to decarboxylate after extraction. Using liquid chromatography and mass spectrometry (LC/MS) to isolate and separate cannabinoids, it is possible to also decarboxylate the material simultaneously in a quick manner.

The required time and temperatures required for each individual cannabinoid in cannabis varies. A common reference point is THCA which requires exposure to a temperature of approximately 220°F/104.44°C for 30-45 minutes to begin decarboxylating. In order for a full decarboxylation to occur, more time is required. For high-CBD hemp, temperatures used may be higher and use a longer exposure. The goal is always to minimize excess heat to reduce damage to the valuable cannabis compounds.
In addition to the cannabinoids, the terpenes contained in the plant must be considered when applying heat. Ideally, the minimum amount of temperature should be used over a longer period of time in order to preserve and not burn off the terpenes. If exposed to high heat, these terpenes can burn, leaving behind undesirable smells and tastes and destroying these important compounds.
Temperatures that exceed 300 degrees F can compromise the integrity of the cannabinoids and terpenes, which is why temperatures in the 200's are recommended.
When a cannabinoid is found in its raw acid form, the mass will be slightly greater than the decarboxylated version. This is important to consider when looking at a potency screening. The mass of THCA or CBDA will be reduced by 12.3% when heated due to the loss of the carbon dioxide molecule. You can easily measure the resulting potency of the resulting THC or CBD by multiplying the acid-form by 0.877.
It is important to note that during the decarboxylation process, not 100% of the 'raw' cannabinoids will be converted. Some small percentage of the raw cannabinoids may still remain after decarbing. This is why you may see some small trace amounts of acid cannabinoids like CBDA on lab tests.
While these heated molecules are favored in most cases, those looking for specific benefits may wish to seek out the natural or 'raw' forms of these cannabinoids. The acid-based forms are not well studied, yet preliminary research points to their ability to provide unique benefits when compared to their activated counterparts. For example, CBDA has been shown via studies to be a COX-2 inhibitor similar to how some NSAIDs function.
Some CBD companies offer raw products which are created by cold-pressing hemp. These products are often combined with activated hemp extracts. One example is a blended CBD + CBDA combination, often found in a 1:1 ratio.
Can high cbd hemp Thca Decarboxylate into delta 9 thc and if so is at the 200 degree temp?.
Hello Leo - the THCA will decarboxylate into Delta-9 THC, yes. This happens at approximately 220°F/104.44°C and requires some exposure time at this temperature to fully decarb. For more information see our article on cannabis compounds.
Hello,
Sorry, I can’t figure out how to write a separate reply without jumping on an existing conversation!
When it comes to vaping or heating CBD for cooking in general, does heating CBD Isolate oils/full spectrum finished products to a high temperature (though still under 200’’C) fundamentally deactivate the compound? As it’s already been decarboxylated during the extraction process?
Slightly confused on this one.
Hope the question makes sense.
Thanks
Hi Storm - heating CBD and other cannabinoids when cooking shouldn't be an issue as long as you're staying well under the boiling point of the compound. For example, the boiling point of CBD is 180˚C/356˚F so if you're under this threshold (I am guessing you meant 200 F not C) then you should be fine!
Hello,
You have noted that decarboxylation will begin after about 30 - 45 minutes when plant is exposed to the temperature of 220 F (105 C). But can you give a reference number for the duration needed for full decarboxylation? Can it be done too much so that the THC or CBD degrades?
Thanks!
Hey Vixxer, there is no black and white procedure to achieve full and complete decarboxylation. The guideline provided is the best general guidance and is widely accepted as being a satisfactory process for decarbing. It could come down to an exact science comparing time and temperature exposures with the test results of the end product - but for any at-home decarbing, this isn't really applicable. It's advised to stay in the 200's as exposure to temperatures at 300 F+ can begin degrading the cannabinoids and terpenes.
Hello, I have a question. Is there a way or a machine to lower the % of THC present in the flowers. I will make an example, have a flower with high CBD and that is 0.8% THC and I want to bring it to 0,2% THC so that it can be sold as CBD. Is there a way to do this? Is decarboxylation involved? Is there a machine that can do this process? I have searched online but I havent found anything but I heard that this kind of process exists.
Thank you in advance.
Hey Tommy - I don't believe there is any machine that can accomplish this at the flower level. There are processes like chromatography and distillation however that can be used to remove/isolate compounds in the final extract.
Hi,
Have a question...
If I cold press hemp and want to activate the CBD, when would be a better time to decarboxylate? Before pressing or after? I'm not finding many resources on cold pressing hemp. Hopefully you can help. Thank you!
Hey Erica - the general consensus with normal extraction is that decarboxylating before extraction is preferred. Decarboxylating the material first will remove moisture content reducing extraction times and increasing yields. That being said cold pressing could be a different beast. Unfortunately, we don't have a definitive answer for you as our products use CO2 and ethanol extraction, not cold pressing.